Preparing chlorates
From PyroGuide
|
Chlorates and perchlorates are among the most often used oxidisers in pyrotechnics. Especially potassium perchlorate (KClO4), potassium chlorate (KClO3), ammonium perchlorate (NH4ClO4), and barium chlorate (Ba(ClO3)2) are used extensively. Chlorates and perchlorates are chemically related but from a pyrotechnical standpoint perchlorates are generally safer to use. Chlorates require special handling and mixtures made with chlorates tend to be more sensitive than similar mixtures based on perchlorates and some may ignite spontaneously. Potassium chlorate is extensively used commercially since it is cheaper than potassium perchlorate, and many accidents have happened that might have been prevented if the corresponding perchlorate was used instead. The use of chlorates is therefore not recommended. If you do decide to use them, make sure you really understand what you are getting involved with.
The amateur pyrotechnist can prepare chlorates and perchlorates at home with some effort. Home made chemicals are usually more expensive than commercially produced chemicals and it is of course laborious, but taking shipping costs into account, the fact that perchlorates and chlorates are sometimes quite hard to obtain in most countries and their usefulness in pyrotechnics it may pay to try to make your own. Chlorates are the easiest to prepare, but again, the use of chlorates is not recommended for amateur use.
Introduction
On an industrial scale, chlorates are prepared by electrolysis. Electrolysing a solution of a chloride at elevated temperatures yields a chlorate. This method can be down scaled quite easily for amateur pyro purposes. Other methods of chlorate manufacture exist that may be of interest for small scale use. They are usually less efficient but the economy of the process is not as important for amateur pyro purposes as it is for industrial set-ups. A second method for example consists of heating a solution of hypochlorite. Sodium and calcium hypochlorite are both quite easily available as bleach and pool chlorinating agent respectively. Upon heating, the hypochlorite will decompose into both chloride and chlorate. The chlorate is separated and purified. Although slow and laborious, the method is simple and requires very little equipment. In the past chlorates were produced even on an industrial scale by bubbling chlorine gas through a hot hydroxide solution. This process is not very well suited for amateurs since chlorine gas is very dangerous to handle. The process is also extremely inefficient, for which reason it was abandoned quite soon after the electrochemical method became feasible at industrial scale.
Electrolytic preparation
The electrolysis is carried out in a diaphragm less cell, containing a solution of a chloride. Several chlorides may be used, but the use of sodium chloride has many advantages. Sodium chlorate is easily converted to a number of other chlorates by metathesis reactions. The most commonly used chlorates in pyrotechnics, potassium and barium chlorate, can both be made in this manner. Potassium chloride and barium chloride may also be used to obtain the respective chlorates directly, but this has many disadvantages as will be discussed below. Only sodium chlorate can be used in the manufacture of perchlorates, due to its high solubility.
Ammonium chloride should never be used, and should in fact not even be present in the cells in trace amounts. It could result in the formation of two dangerously sensitive and explosive compounds, nitrogen trichloride (NCl3) and ammonium chlorate (NH4ClO3). The formation of both of these compounds should be avoided at all times. Not only can they explode by themselves when present in significant quantities, they can also lead to spontaneous ignition of pyrotechnic mixtures contaminated with even small amounts.
Theory
Mechanism of chlorate formation
The reactions taking place in chlorate cells are not fully understood even today. The theory of Foerster and Mueller regarding the reactions in chlorate cells, developed about 80 years ago, is the most accepted. The following reactions are said to take place at the electrodes:
At the anode:
At the cathode:
The dissolved chlorine gas can then react with water to give hypochlorous acid:
From this reaction it can be seen that if the chlorine does not dissolve but escapes to the atmosphere, no H+ will be generated to neutralise the OH- formed at the cathode and the pH of the electrolyte will increase.
The hypochlorous acid thus formed will react in acid-base equilibrium reactions with water to give hypochlorite ions and chlorine gas (dissolved). The exact concentrations of dissolved Cl2, ClO- and HClO depend on the pH, temperature and pressure among other things. In the solution, chlorate will be formed (mainly) by the following reactions:
andThese reactions take place at a rather slow rate. Since this reaction pathway is the most efficient one as we will shortly come to see, the conditions in the cell are usually optimised to increase their reaction rate. The pH is kept within a range where HClO and ClO- are simultaneously at their maximum concentration (which is at around pH=6). The temperature is kept between 60 and 80 degrees centigrade, which is a good compromise between the temperatures required for a high reaction rate, low anode and cell body corrosion and high chlorine solubility (remember the chlorine evolved at the anode has to dissolve in the solution to start with). Many cells also have a large storage tank for electrolyte in which the electrolyte is kept for a while to give these reactions some time to take place.
Alternatively, chlorate may also be formed by oxidation of hypochlorite at the anode as follows:
Oxygen is evolved in this reaction, which means a loss of current efficiency (the energy used for oxidising the oxygen in water to the free element is lost when the oxygen escapes to the atmosphere). When the reaction routes are worked out, it turns out that following this path 9 faradays of charge are required to produce 1 mole of chlorate, whereas only 6 faradays are required to do that following the route mentioned earlier. Therefore, optimising the conditions for that route improves current efficiency.
To prevent the products from being reduced at the cathode again, a membrane around the cathode was employed in the past. Today, small amounts of chromates or dichromates are added. A layer of hydrated oxides of chromium will then form around the cathode effectively preventing hypochlorite and chlorate ions from reaching the cathode surface.
Finally, it should be mentioned that the reactions forming perchlorates do not take place until the chloride concentration has dropped to below about 10%. Therefore, cells can be constructed and operated in such a way that chlorate is produced almost exclusively. The chlorate can then be purified and fed into a perchlorate cell. Depending on the type of anodes used in the chlorate cell, the purification step may also be skipped and the electrolysis continued until all chloride has been converted into perchlorate. Although slightly less efficient (and therefore not used a lot in industrial set-ups), this is much less laborious and therefore probably the preferred method for home set-ups.
Cell voltage
The current through a cell is related to the reaction rate. Therefore, to obtain a constant reaction rate that suits the cell design, a constant current is usually employed. The voltage over the cell will then fluctuate depending on conditions and cell design. The power consumed by the cell is the product of current and voltage, according to equation P = I * V. From that it can be seen that reducing the voltage over the cell results in a lower power consumption, an important fact for industrial operations. The factors influencing the cell voltage have been thoroughly investigated. Most important are the anode - cathode spacing, the concentration of the electrolyte, the surface area and materials of the electrodes, the temperature and the pH. Without going into details, the cell voltage usually lies in the range 3.5 - 4.5 volts. Of this, approximately 3 volts are required to get the oxidation of chloride to chlorate to take place (and the hydrogen reduction at the cathode), while the rest is used to overcome the resistance of the cell, according to Ohm's law V=I*R. From this law it can be seen that there are two ways to maintain a constant current through a cell: either the voltage over it may be varied or its resistance may be changed. Adjusting the voltage over a cell to maintain a constant current can be done manually or with an electronic circuit. If the power supply does not allow voltage adjustment (such as old PC power supplies or battery chargers for example) or the required electronics are not available, adjusting the resistance of the cell is another option. This could in principle be done by adjusting each of the factors mentioned earlier, the most practical of which is probably the anode-cathode distance. By increasing the distance between the electrodes the resistance of the cell is increased, which reduces the current through the cell. One thing to keep in mind when doing this is that it with decreasing resistance, the heat generated in the cell is increased. Depending on the anode material used it may then be necessary to cool the cell to prevent excessive erosion, more on that later.
Cell construction
Cells can range in complexity from a glass jar with a nail and a old battery electrode to well designed, corrosion resistant cells with thermostats, pH control, circulating electrolyte and coulometers. Even the simplest of cells will work, but it will require more maintenance. If the chlorates are going to be prepared on a more or less regular basis, it probably pays to spend some more time designing a cell. It will also improve efficiency somewhat, but unlike in industrial set-ups where high efficiency is mandatory to be able to compete, the home experimenter can do with less efficient cells. The two main disadvantages of a low efficiency is that it takes more time for the conversion to complete, and that more electricity is required. To give some indication of the power consumption of the process: typical figures for industrial cells lie in the range 4.5 to 5.5 kWh per kg of sodium chlorate.
In this section some of the things to consider when building and designing chlorate cells will be discussed. The reader can design his own cell based on the information given. An example of a cell, the small test cell I currently use to experiment with, has been given but it is by no means perfect, and it is probably better to design your own. The example has merely been given to illustrate some principles.
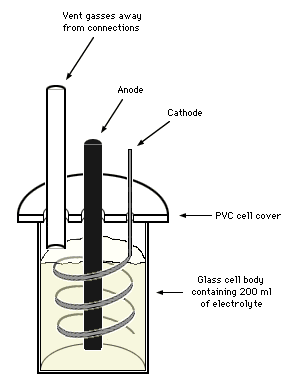
The example given here consists of a small cell, of 200 ml electrolyte volume. The cell is normally operated with graphite or graphite substrate lead dioxide anodes. Platinum sheet has also been tried with, unsurprisingly, good success. The electrolyte consists of sodium chloride with either some potassium dichromate or potassium fluoride added, depending on whether graphite or lead dioxide anodes are used. The cathode consists of a stainless steel wire spiralling down. The wire is corroded where it is not submerged, so it has to be replaced occasionally. The connections to the anode and cathode are made outside the cell but do corrode from the gasses and electrolyte mist. This is partially prevented by leading the gasses away from the connections with a vent tube, as shown in the picture. Covering the connections with hot melt glue also helps, but the heat generated in a faulty connection may cause the hot melt to melt.. The temperature is controlled by placing the cell in a water bath, which acts as a heat sink. If the temperature is too low, styrofoam isolation is provided. The cell is operated outside, causing the temperature to fluctuate between day and night. The pH is checked about twice a day and adjusted if necessary with hydrochloric acid. The power source used is an old computer power supply. The output voltage can be regulated within certain limits and this is done to maintain a current of about 4 amperes. An other model computer power supply was used previously that did not allow control over the output voltage. Current adjustment was done by widening or narrowing the cathode spiral, effectively reducing or increasing the anode-cathode distance.
Theoretically, if 100% efficiency could be reached, the cell would have the capacity to convert approximately 35 grams of sodium chloride to 64 grams of sodium chlorate per day. Using a metathesis reaction with potassium chloride this would yield 74g of potassium chlorate. In practice the average yield is about 40 grams of potassium chlorate a day from which an efficiency of 55% can be calculated.
Cell volume
This is the main factor affecting a cells capacity, provided the power supply can provide the necessary current. As a rule of thumb no more than 2 amperes per 100 ml of electrolyte must be passed through a chlorate cell. Under more optimal conditions a higher amperage may be tolerable, still maintaining reasonable efficiency whereas in less optimal conditions 2 amperes may be too high and a lot of chlorine will be lost, leading to lower efficiency and rising pH. A current of 2 amperes will convert approximately 0.73 gram of sodium chloride to 1.32g of sodium chlorate per hour (assuming 100% efficiency). After extracting, metathesis reactions and recrystallising that will yield 1.53 g potassium chlorate. So, for example, to produce 100 grams of potassium chlorate a day at least 100 grams / 1.53 grams / 24 hours * 100 ml = 272 ml of electrolyte is required. To maintain that rate of conversion the cell will then require 272 ml / 100 ml * 2 amperes = 5.44 amperes. If a cell is less efficient than 100%, which every cell is, increase these figures proportionally (so at 50% efficiency: 100% / 50 % * 272 ml = 544 ml of electrolyte, consuming 10.88 amperes of current to maintain the same rate of production). The example cell described above contains 200 ml of electrolyte. Thus, it should be operated at a current of 4 amperes, and the maximum daily yield is 100/272 * 200 = 74 g of potassium chlorate after processing the electrolyte. These figures were also mentioned in the cell description without explanation.
Cell body materials
One of the main problems in chlorate cells is the corrosiveness of the electrolyte. Only very few materials do not corrode when in contact with the electrolyte or its fumes. Most metals corrode, many plastics will and even glass does under some circumstances.
Some metals, such as steel, can be used if they are protected from corrosion in some way. For that purpose it can be coated with a resistant material such as teflon or some types of rubber, or it can be 'cathodically protected'. This means means it is used as a cathode. The negative potential prevents the steel from being oxidised if the current density (current per unit of surface area) on the steel is high enough.
Some metals, such as titanium, zirconium, tantalum and niobium, form a protective film when they are in contact with the electrolyte. This prevents them from further corrosion, and they therefore find extensive use in industrial set-ups (particularly titanium because it is the cheapest). For amateurs the difficulties in working with these metals and their high price restricts their use somewhat. In small scale set-ups glass and plastics such as PVC (Poly(Vinyl Chloride)) or PTFE (Poly(TetraChloroEthene)) are more easily available, easier to work with and much cheaper.
Electrode materials
The range of suitable electrode materials is very limited. Especially the anode material is critical. The positive charge on the anode promotes oxidation and the evolving oxygen attacks many anode materials. Several anode materials have been considered over the years. Today's main options are listed below along with a short description.
Anode materials
Graphite: graphite is cheap and easy to obtain. It does however corrode at a comparatively fast rate. This makes it necessary to replace the anodes every so often and to filter the electrolyte before further processing which can be difficult and laborious due to the small size of the carbon particles. Graphite is not suitable for making perchlorates. When the chloride concentration of the electrolyte drops to the point where perchlorate formation begins (about 10% w/v), the graphite begins to oxidise at a great rate, yielding no or only traces of perchlorate. Cells operating with graphite anodes must also be maintained at a relative low temperature to limit anode erosion, which translates to a lower cell capacity. Graphite rods can be found in old manganese dioxide-zinc batteries or in welding shops where they are sold as 'gouging rods'. They can be treated with linseed oil to reduce corrosion. A practical method for the home experimenter has been devised by Rich Weaver, and is well described on Mike Brown's page. Old battery electrodes do not need to be treated with linseed oil.
Platinum: The obvious disadvantage of platinum is its high price. However, platinum anodes corrode only at a very slow rate and are suitable for perchlorate production. They therefore provide an almost ideal anode material. High efficiency can be reached with platinum and processing of the electrolyte is greatly simplified.
Lead dioxide: Lead dioxide provides an economical alternative to platinum. Lead dioxide anodes can be made at home. This takes some work and effort, but the anodes are cheap, fairly resistant to corrosion even at higher temperatures and are suitable for perchlorate production.
Manganese dioxide: Another oxide that is conductive and resistant to oxidation. It is made by thermal decomposition of manganese nitrate pasted onto a substrate. This type of anode seems quite promising for amateur pyro use. For more information, the user is referenced to patents in the literature list below.
DSA: DSA stands for Dimensionally Stable Anode. This is the common term used to refer to anodes consisting of a layer of noble metal oxides (usually RuO2 and TiO2) coated onto a substrate, usually titanium. This type of anode is finding increased use in industrial cells because of its comparatively low cost when compared to platinum and its resistance to corrosion. Some of the chemicals required to manufacture these anodes (particularly RuCl3 and tetra-butyl titanate, Ti(OBu)4) are expensive and perhaps difficult to handle safely. However, if the chemicals can be obtained and suitable equipment is available, the procedure to make the anode seems fairly straightforward and may be an option.
Magnetite: This has found use in industry in the past, but is rarely used nowadays. It corrodes, but not very quickly and it can be used for perchlorate manufacture. The anodes are made by melting and casting FeO.Fe2O3 into the required anode shapes. I have little literature available on this material, so it is not further discussed here.
Cathode materials
Both stainless and mild steel find widespread use as cathode materials. Brass and copper may also be used. Each of these metals is protected to a certain extent by the negative charge present on the cathode as long as they are submerged and the current per surface area is high enough. Unsubmerged parts of the cathode corrode at a high rate however due to the action of evolving gasses and droplets of cell electrolyte.
It seems that under some conditions the chromium in stainless steel can dissolve, even though the cathode does not seem to corrode. A yellow electrolyte is the result from which barium chromate can be precipitated even if no chromate was added, which will be described later. The presence of chromates could lower the efficiency of cells employing lead dioxide anodes.
Finally, contamination of the final product with copper (from brass or copper erosion products) can be dangerous when the product is to be used in pyrotechnic purposes. Although this is unlikely to be a great problem since the impurities are usually removed easily and completely by filtration (as will be described later) it is good to be aware of the possibility.
pH and temperature control
Although not essential for chlorate manufacture, controlling the temperature and pH will increase cell efficiency and therefore the capacity of a cell. Temperature control can be anything from a sophisticated thermostat and heating element (or a cooling element) to simple insulation around the cell or a cold water bath. As mentioned earlier, part of the electric energy is lost as heat in the cell. Small cells operating at high currents can sometimes reach temperatures of 80 to 90 deg C. Though high temperatures will improve efficiency, temperatures as high as that will also increase anode corrosion and it is therefore usually considered better to maintain a temperature in the range 60 to 80 deg C to get the best of both worlds. Graphite anodes tend to erode faster than other types though, especially at higher temperatures, and cells employing these are therefore usually operated at 40 deg C to limit anode erosion.
Like temperature control, pH control is not essential for chlorate manufacture. Efficiency is improved greatly however if the pH is kept within the range 5.5 - 6.5 (slightly acidic) as explained in the theory section. Graphite anodes also tend to erode faster at high pH, so maintaining the pH will extend graphite anode life. In commercial set-ups pH control is done manually by periodic additions of hydrochloric acid. Automated pH control seems to be difficult and expensive to realise. If anyone devices a practical method of doing this, I'd be interested to hear about it.
Preparing the electrolyte
When just starting a first batch of chlorate a fresh electrolyte has to be prepared. When the cell has been operated before, the electrolyte from the previous cell is available to prepare the electrolyte for a new batch. Also, the material left behind from the extraction and purification steps can be added to the next cell as it may contain some residual chlorate. That way no product left in the solution after processing is lost.
As mentioned, it is common to use sodium chloride to prepare sodium chlorate first, which is then converted to potassium or barium chlorate later. Even though both compounds may be prepared directly from potassium or barium chloride, using sodium chloride as a starting material has advantages. Mainly, it makes processing of the electrolyte much easier since sodium chlorate is very soluble. It is therefore easily separated from insoluble impurities which are almost always present. It will be assumed that sodium chloride is used. If for some reason the use of other compounds is desired, the procedure and amounts may need to be adjusted.
Preparing fresh electrolyte
1. Prepare a saturated solution of sodium chloride. Take about 40 grams for every 100 ml of solution and bring the solution to a boil. Then allow to cool to room temperature again. Some sodium chloride will crystallise as the solution cools. The solution is then filtered to obtain a clear saturated solution.
2. Optionally, 2 to 4 g/l of potassium dichromate, potassium chromate, sodium chromate or sodium dichromate may be added to improve efficiency. These compounds are suspected carcinogens, so if you choose to add any, know the hazards involved and act accordingly. If lead dioxide anodes are used, do not add potassium dichromate as it will only reduce efficiency. Instead, 2 to 4 g/l of sodium or potassium fluoride may be used. Although not carcinogenic, the fluorides are nasty compounds as well and should be handled properly.
3. Finally, the pH of the solution can be adjusted. A pH of around 6 is optimal, but anything between 5.5 and 6.5 is reasonable. The pH can be increased by addition of sodium hydroxide solution and it can be decreased by adding hydrochloric acid. Do not use too concentrated solutions for adjusting the pH. A concentration of 2% (w/v) for both solutions is convenient to work with.
Recycling old electrolyte
When electrolyte from a previous batch of chlorate is available the following steps can be used to recycle the electrolyte.
1. If the electrolyte is not clear but has solid particles in it, filter to remove these. See the section on filtering below.
2. dissolve any impure chlorate from the purification and extraction steps.
3. Now, re-saturate the solution with sodium chloride. The procedure mentioned above in step 1 of 'preparing a fresh electrolyte' may be used.
4. The chromate, dichromate or fluoride if added is still present so does not need to be replenished. The pH should be readjusted, like in step 3 for preparing a fresh solution above.
Operating the cell
Voltage and current
As explained in the theory section the voltage over the cell may vary. The current should be kept more or less constant at a value determined by the cell design. As a rule of thumb, supply 2 amperes of current per 100 ml of electrolyte. If graphite anodes are used it is better to supply less current since that will increase anode life (30 mA per square centimetre of anode surface area is typical). A constant current supply is of course the most convenient to use for regulating current, but manually adjusting the voltage from time to time also works well. The current usually only changes very gradually, and the precise value is not very critical. In any case, measure the current at regular intervals and record them. That information is required to determine when a batch is complete, as described below in the paragraph 'running times'.
Maintaining optimal conditions
As explained, the pH of the electrolyte will tend to rise. Also, some of the water will evaporate and some will be consumed in the reaction. The temperature may also vary with ambient temperatures. For good efficiency these variables must be kept within certain limits.
Every once in a while, make up for evaporated and consumed water. This can be done with water, but it is better to use a saturated sodium or potassium chloride solution. That way, the chloride concentration will be kept at a higher level which improves efficiency. It will also prevent excessive formation of perchlorate, and in graphite anode based cells it reduces anode wear.
The pH will rise during operation of the cell, and it is best to lower the pH regularly to a value of about 6. A high pH value is best corrected by adding hydrochloric acid occasionally. If too much HCl is added, it may be corrected with sodium hydroxide solution. The pH is self correcting to a certain extend as well, since at very low pH the cell will produce more chlorine gas. This will then escape, raising the pH again. Measuring the pH of the solution can be done with common pH paper. However, if the paper is simply dipped directly into the electrolyte the hypochlorite present will usually bleach the paper making a measurement impossible. This problem can be overcome by boiling a sample of the solution for 5 minutes and measuring the pH of that. The boiling destroys the hypochlorite.
The temperature will usually reach a more or less constant value quickly. If the cell is placed outside, the temperature may vary between night and day and between seasons of course, so then some sort of control may be necessary to maintain the optimum. Usually, cooling is necessary but it will depend on the specific cell design as explained in the theory section.
Running times
The time required to convert a certain amount of chloride to chlorate depends on the current and the efficiency. The latter can vary dramatically from cell to cell and it is therefore not possible to state precise running times. It is however possible to calculate the required running times for a hypothetical cell operating with 100% current efficiency. The calculation will show that the conversion of one mole of chloride to chlorate requires 160.8 amperage hours. So, for example, a cell containing 100 grams of sodium chloride will require 100/58.6 * 160.8 = 274.4 Ah if it operates at 100% efficiency. For a current efficiency other than 100%, increase the running times in proportion (to convert 100g of NaCl with 80% current efficiency one needs 274.4/80*100 = 343 Ah). So, if a current of 3 amperes flows through the cell, it requires 274.4 / 3 = 91.47 hours (91 hours, 28 minutes) to finish.
Processing the electrolyte
When done, the product must be extracted from the electrolyte and the electrolyte can be recycled for the next batch.
Filtering
The electrolyte usually contains suspended solid particles, even though they are not always visible. Suspended particles can be detected with the use of the Tyndall effect. Shine a bright flash light through the side of a glass container containing the solution. If no suspended particles are present the light beam cannot be seen going through the solution. If suspended particles are present they will scatter the light and make the beam visible.
Usually, the impurities consist of erosion products of the anodes, the cell walls, and the unsubmerged parts of the cathodes. These particles may be very small and are not always easily removed with common filtering paper. Filtering through a layer of diatomeous earth (sold in shops for aquarium supplies) in a filter or on a piece of cloth sometimes solves the problem. Another great idea for a filter comes from E.S. However, just filtering will not always remove all solid impurities. A common impurity that is hard to remove is suspended iron hydroxide, originating from corrosion of (stainless) steel cathodes. The fluffy, voluminous form of the material often gives it a white or yellowish foggy appearance. This is next to impossible to remove unless some sodium hydroxide or pool coagulant is added first. This causes the iron hydroxide particles to coagulate, making them easy to remove by filtration. Another possibility is to add hydrochloric acid to lower the pH to between 2 and 3. This will dissolve the iron hydroxide. If sodium hydroxide is then added to raise the pH to above 7 again, the iron hydroxide is precipitated in a more dense form which is easily removed by filtration, even with common filter paper.
In this step, the advantages of using sodium chloride will become evident. When potassium chloride is used instead potassium chlorate crystallises during operation of the cell due to its relatively low solubility. To separate the potassium chlorate from insoluble impurities the electrolyte has to be filtered hot. The solution usually takes quite a long time to pass through the filter and if it cools during this time, potassium chlorate will crystallise and block the filter. Alternative methods have been developed to separate potassium chlorate from insoluble impurities. For example, the solution may be boiled and sufficient water added to dissolve all potassium chlorate. If the solution is then allowed to cool slowly, crystals of potassium chlorate will form on the suspended insoluble impurities. These will sink to the bottom, usually leaving a clear solution. The clear solution is then carefully decanted and allowed to cool further . This method will not remove the insoluble purities as well as filtering will but it is much less laborious.
Destruction of hypochlorite
Next, the electrolyte is boiled to decompose remaining hypochlorite. 15 minutes of vigorous boiling is sufficient. After that, the pH of the solution is checked and it is made slightly alkaline by adding sodium hydroxide solution. Bring the pH to between 8 and 9.
Metathesis reaction
At this point, a clear solution of sodium chlorate (with residual chloride) has been obtained. This can be used either to prepare potassium or barium chlorate (or other chlorates which are not further elaborated upon here), or it can be used to prepare perchlorates, described elsewhere.
Potassium chlorate is by far the most commonly used chlorate in pyrotechnics. For practical purposes, the preparation of this compound is discussed here. For the preparation of barium chlorate the amounts will have to be adjusted.
1. Weigh out either 127g of potassium chloride or 355g barium chloride for every 100 g of sodium chloride that was started with, depending on whether you want to prepare potassium or barium chlorate. Dissolve this in as little water as possible (dissolve in minimum amount of boiling hot water, add a bit more water and allow to cool. Nothing should precipitate. If it does, add some more water and heat again)
2. Add this solution to the electrolyte. A white precipitate of potassium or barium chlorate should form.
3. Bring the solution to a boil. Add 20 ml amounts of water to the solution in 5 minute intervals until all chlorate has dissolved. If all chlorate dissolves upon heating without the addition of extra water, allow the water to evaporate until a thin crust of chlorate forms on the surface (indicating that the saturation point has been reached). Then add 20 ml of water and boil for a minute to re dissolve the crust.
4. Allow the solution to cool to room temperature. Potassium or barium chlorate will crystallise. If it has cooled to room temperature, cool further to 0 deg C.
5. Filter to obtain the crude chlorate crystals. Rinse them thoroughly with ice-cold water. The filtrate can be saved to prepare the next electrolyte, as is described in the section on recycling electrolyte.
Purifying the product
The crude product can be purified by recrystallisation. The low solubility of potassium chlorate makes this method very convenient to use and will greatly improve the purity with a relatively small loss of product. Barium chlorate is somewhat more soluble and to prevent losses it is a good idea to use the impure barium chlorate 'waste' from this procedure in the electrolyte of a new cell. Some treatment is necessary, which was described earlier. If a single recrystallisation step does not yield a sufficiently pure product, the method can be repeated to further increase the purity. Usually one or two recrystallisations will yield a product that does not impart the characteristic yellow color of sodium impurities to a flame.
1. Place the crude product in a pan and add 100 ml of water for every 35g of crude potassium chlorate or 50g of barium chlorate. Bring this to a boil.
2. Add 20 ml amounts of water to the boiling solution until all the chlorate has dissolved.
3. Check the pH of the boiling solution. It should be neutral or slightly alkaline. If it is acidic, add potassium hydroxide solution until it is slightly alkaline (pH 7) again. If this is not done, traces of acid may be included in the product making it very dangerous to use in pyrotechnic compositions.
4. Allow the solution to cool to room temperature. The chlorate will crystallise.
5. Filter and rinse the crystals in the filter well with ice cold water. The filtrate may be used to prepare the electrolyte for a new cell, as was described in the section on recycling old electrolyte.
6. The crystals may be dried in an oven at 100 deg C.