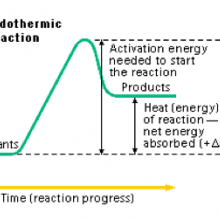
An endothermic reaction is one in which the products have more energy than the reactants, and thus a net input of energy, usually in the form of heat, is required. Endothermic reactions are often described as reactions that "feel cold", and contrast with exothermic reactions, in which heat is released.
Although the process of bond breaking amongst reactants in a chemical process requires an initial input of energy (the activation energy), in the case of an endothermic reaction, the energy released when bonds are formed to create reactants is less than that required to break the bonds in the products; bonding electrons in the products are therefore at a higher energy than the reactants. Heat energy from the material surrounding the reactants is usually what breaks their bonds, so as heat energy is transferred from the surroundings to the reactants, the surroundings get colder. This is often summarized in a chemical equation as follows:
- Reactants + Energy → Products